
The impacts of hydrate mining
Fear of disasters
In recent years the potentially negative impact of meth-ane hydrate mining on the marine environment and climate has been a source of heated debate in professional circles. Concerns have been voiced that extracting the hydrates could release vast quantities of methane into the atmosphere. In this event the consequences would be disastrous, as methane is a greenhouse gas about 20 times more potent than carbon dioxide. Some scientists have claimed that such an increased release of methane from the oceans could accelerate climate change.
The possibility that hydrate mining could generate submarine landslides on steep continental slopes has also been discussed. Like avalanches in mountainous regions, submarine landslides are natural events. They occur on continental margins where thick layers of soft sediment have accumulated, such as near river mouths. Similar to the alpine snow, the sediment at some stage becomes so heavy that it begins to slide downhill. Gas hydrates cement the pores between the fine particles of sediment and thus stabilize the seabed. Some scientists have claimed that dissociating the methane hydrates would destabilize the sea floor, and in the worst case scenario huge packages of sediments could slide downhill, triggering powerful tsunamis along coastal areas.
- 3.14 > In freshwater lakes, such as this one near Fairbanks in Alaska, methane bubbles can freeze in ice.

Environmental damage from hydrate mining?
It is not uncommon for slopes to slide. There is even scientific evidence that landslides have been responsible for severe tsunamis. One such example is the Storegga Slide which occurred off the coast of Norway 7000 years ago, when a large section of the Norwegian continental slope collapsed and sank. The motion was so great that 20-metre waves struck the shores of Scotland. This incident had nothing to do with methane hydrates, however. The Storegga slope began to move because, after the Ice Age, the Scandinavian continental plate began to slowly rise, causing a portion of the slope to break off. Such major slides are very rare, only striking every few thousand years.
Smaller landslides, on the other hand, are very common. A certain number of slopes around the world have sufficient accumulation of sediment for even a small disruption to generate a slump. For this reason it is vital that any potential drilling site be closely evaluated in advance. Scientists assert that environmental impact assessments in future will evaluate the risk of land-slides before methane hydrate mining can begin. However, uniform standards governing the survey of methane hydrate areas have yet to be developed. Japan and Korea, who are leading the way in this field, will for the time being choose shallow marine areas such as ocean basins for their activities, in order to largely preclude the risk of landslides.
Relatively small-scale methane hydrate mining does not cause landslides or trigger tsunamis. Moreover, the investment costs are so exorbitant that participating companies are unwilling to take the risk of their drilling equipment being destroyed on the seabed.
The introduction of carbon dioxide reduces the risk of landslides from hydrate mining. Carbon dioxide is injected into the hydrate to replace the methane being released. The CO2 itself reacts with the water to form a solid hydrate, which re-stabilizes the sediments.
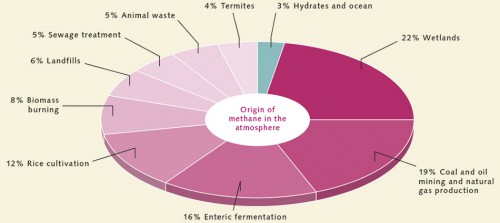
- 3.15 > Atmospheric methane comes mainly from land-based sources – particularly wetland areas. Only a small proportion is contributed by marine methane hydrates. Even global warming will not substantially increase this amount.
Point source disruption
In one other respect, too, experts now tend to consider methane hydrate mining as relatively benign. Unlike the mining of massive sulphides and manganese nodules, the disruption of fragile seabed habitats is isolated, because no major mass movement is involved. The sediment is churned up only in the immediate vicinity of the drilling site.
Even where several boreholes are drilled during the development stage of a reservoir, any disruption is relatively minor. Oil and gas industry experience shows that drilling does not affect the marine environment to any measurable extent – apart from disasters of the magnitude of the Deepwater Horizon oil platform in the Gulf of Mexico.
Greenhouse effect
Water vapour, carbon dioxide (CO₂), methane and other trace gases in the atmosphere at first allow the sun’s energy (short-wave radiation) to pass through to the Earth. This energy is converted to heat on the Earth’s surface and a large proportion is emitted back into the atmosphere in the form of long-wave radiation. Like the glass windows of a greenhouse, however, the gases prevent the long-wave thermal radiation from escaping into space. The Earth heats up.Could methane reach the atmosphere?
The notion that large quantities of methane can flow up out of the oceans is not new. Some people even believed that this was the reason behind the mysterious disappearance of ships in the Bermuda Triangle. According to this theory, enormous bubbles of methane rose from the depths and swamped the ships. We now know that such large bubbles cannot break loose from hydrates. Nor will hydrate mining cause significant amounts of methane gas to rise freely into the atmosphere. There are several reasons for this:- Scientists recommend the mining of only methane hydrate deposits which are covered by a layer of sediment at least 100 metres thick. This amount of sediment prevents any methane bubbles which may form in the vicinity of the borehole from being released into the water.
- Unlike natural gas and oil, methane does not shoot up out of the borehole on its own. The hydrate must gradually break down (dissociate), resulting in the slow release of the methane. There is therefore no danger of a blow-out similar to the Deepwater Horizon oil platform in 2010. No large volumes of meth-ane gas will be released to flow to the surface.
Does global warming accelerate the breakdown of methane hydrates?
Methane is a powerful greenhouse gas, and we understand that it is responsible for 15 per cent of the greenhouse effect. For this reason scientists have in recent years tried to estimate how much methane is released into the atmosphere annually. Wetlands, where large volumes of dead plant material are broken down by methane-producing bacteria, are considered to be the main source. Other sources include the stomachs of cows and other ruminants, rice cultivation, and oil and gas production. How much methane, if any, will in future be released into the atmosphere as a result of global warming has long been the subject of controversial debate. Scientists base their calculations on 4 different types of methane hydrate reservoir types: IN ONSHORE PERMAFROST REGIONS: Such methane hydrate reservoirs are found in regions such as Alaska, Canada and Siberia. They contain only approximately 1 per cent of the global volume. Their impact on the climate would be equally insignificant. In most of these regions the deposits are situated at depths of more than 300 metres. Scientists believe that global warming would, at most, cause the upper layers of methane hydrate to thaw. This process is likely to take several thousand years. Deposits at depths of about 20 metres would be much more sensitive to warming, but these are rarer and the total volume of methane is minimal. IN FLOODED PERMAFROST REGIONS ON THE ARCTIC SHELF: Rising sea levels after the last ice age were responsible for the flooding of permafrost regions in the Arctic. Because the temperature of the water, being slightly above 0 degrees Celsius, is considerably warmer than that of the Arctic air, the flooded permafrost began to thaw. Now, several thousand years later, the thaw has reached the depth of the gas hydrate stability zone (GHSZ). The hydrates are slowly dissociating and releasing methane. This is occurring in many parts of the seabed, including the Siberian shelf. The impact of human-induced climate warming on this process will continue to be minimal. Computer models show that if any methane hydrates do in fact thaw, this process will be limited to those buried in sediments at depths of only 10 to 20 metres. Such deposits are, however, rare. As the water in the shelf regions is relatively shallow, this methane would indeed be released into the atmosphere. It is estimated that the flooded permafrost regions of the Arctic shelf contain less than 1 per cent of the global volume of methane hydrates. ON CONTINENTAL MARGINS (UPPER BOUNDARIES OF THE GHSZ): These methane hydrate deposits are situated exactly where the GHSZ begins – mostly at depths of 300 to 500 metres. Because of their location at the upper boundary of the GHSZ they are particularly vulnerable to ocean warming. Even minimal warming would cause them to start to dissociate. In other regions gas hydrates act as a type of plug and obstruct deeper-lying methane gas bubbles. These plugs could also break loose to release additional methane gas. It is estimated that deposits along the continental margins and the upper margins of the GHSZ contain about 3 per cent of the global volume of methane hydrates. ON DEEP-SEA CONTINENTAL MARGINS: The largest proportion, about 95 per cent, of methane hydrate deposits is found in the sediments of deep-sea continental margins at depths of 500 to 3000 metres, where water pressures are high. Rising seawater temperatures as a result of climate change have little effect on the stability of these hydrates. Firstly, the pressures are so high that a minimal temperature increase is not enough for the hydrates to break down. Secondly, it will take many thousands of years for the warming to spread from the surface to the deep water or the sediment.
- 3.17 > Deposits of methane hydrates are found in different settings around the world. The effects of climate change and global warming are not the same for each setting. Most methane hydrates are buried beneath the deep ocean, where they are largely protected from dissociation.
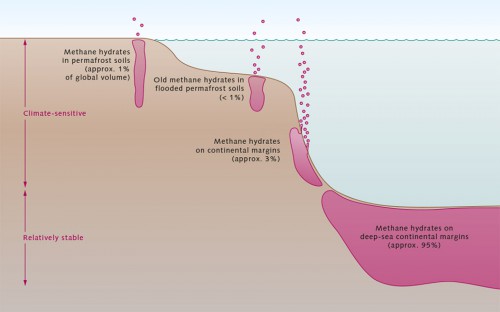
- Because many marine areas have not yet been adequately explored, it is impossible to say with any certainty what the exact proportional distribution is. Most scientists, however, agree that climate change will not trigger any catastrophic mass meltdown of methane hydrates, because by far the greatest hydrate volumes are stored in the sediments of deep-sea continental margins. One topic of discussion, however, is whether methane has ever before been released en masse from hydrates.
Apparently climate warming was responsible for periodic mass meltdowns millions of years ago. These then started a chain reaction and the methane gas is said to have heated the Earth even more. Some researchers believe that this could have been the case with the Paleocene-Eocene Thermal Maximum (PETM) roughly 55 million years ago. Within a period of 20,000 years during the PETM, worldwide temperatures rose by an average of 6 degrees Celsius. This is a great deal when we consider that climate researchers today anticipate significant changes to the climate from a global temperature rise of little more than 2 degrees Celsius. The causes of the Paleocene-Eocene Thermal Maximum remain a source of controversial debate among scientists, and some suspect that it could have been triggered or at least intensified by the release of methane gas.
Experts believe that the dissociation of gas hydrates will contribute little to global warming during the next few centuries. But if we look at longer periods of time, through several millennia, it is certainly possible for increased quantities of methane to be emitted. Initially the human-induced, anthropogenic carbon dioxide emissions would lead to an extended period of warming, as most of the carbon dioxide released would still be present in the atmosphere in more than a thousand years – long after we are supplying our energy needs from renewable sources. Such long-term warming would cause the hydrates to slowly break down. It is therefore not inconceivable that the long-term effect of today’s carbon dioxide emissions could intensify the dissociation of gas hydrates, adding further momentum to the greenhouse effect.