
Oxygen in the ocean
Oxygen – product and elixir of life
Carbon dioxide, which occurs in relatively small amounts in the atmosphere, is both a crucial substance for plants, and a climate-threatening gas. Oxygen, on the other hand, is not only a major component of the atmosphere, it is also the most abundant chemical element on Earth. The emergence of oxygen in the atmosphere is the result of a biological success model, photosynthesis, which helps plants and bacteria to convert inorganic materials such as carbon dioxide and water to biomass. Oxygen was, and continues to be generated by this process. The biomass produced is, for its part, the nutritional foundation for consumers, either bacteria, animals or humans. These consumers cannot draw their required energy from sunlight as the plants do, rather they have to obtain it by burning biomass, a process that consumes oxygen. Atmospheric oxygen on our planet is thus a product, as well as the elixir of life.
- 2.13 > Marine animals react in different ways to oxygen deficiency. Many species of snails, for instance, can tolerate lower O2 levels than fish or crabs. The diagram shows the concentration at which half of the animals die under experimental conditions. The average value is shown as a red line for each animal group. The bars show the full spectrum: some crustaceans can tolerate much lower O2 concentrations than others. /dt>

Oxygen budget for the world ocean
Just like on the land, there are also photosynthetically active plants and bacteria in the ocean, the primary producers. Annually, they generate about the same amount of oxygen and fix as much carbon as all the land plants together. This is quite amazing. After all, the total living biomass in the ocean is only about one two-hundredth of that in the land plants. This means that primary producers in the ocean are around two hundred times more productive than land plants with respect to their mass. This reflects the high productivity of single-celled algae, which contain very little inactive biomass such as, for example, the heartwood in tree trunks.
- 2.14 > Oxygen from the atmosphere enters the near-surface waters of the ocean. This upper layer is well mixed, and is thus in chemical equilibrium with the atmosphere and rich in O2. It ends abruptly at the pyncnocline, which acts like a barrier. The oxygenrich water in the surface zone does not mix readily with deeper water layers. Oxygen essentially only enters the deeper ocean by the motion of water currents, especially with the formation of deep and intermediate waters in the polarregions. In the inner ocean, marine organisms consume oxygen. This creates a very sensitive equilibrium.
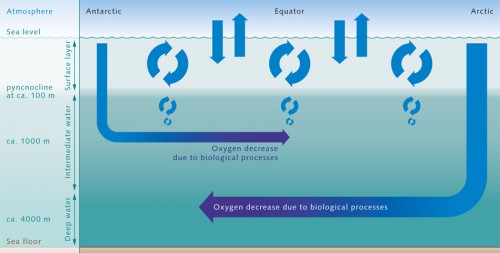
- Photosynthetic production of oxygen is limited, however, to the uppermost, sunlit layer of the ocean. This only extends to a depth of around 100 metres and, because of the stable density layering of the ocean, it is largely separated from the enormous underlying volume of the deeper ocean. Moreover, most of the oxygen generated by the primary producers escapes into the atmosphere within a short time, and thus does not contribute to oxygen enrichment in the deep water column. This is because the near-surface water, which extends down to around 100 metres, is typically saturated with oxygen by the supply from the atmosphere, and thus cannot store additional oxygen from biological production. In the inner ocean, on the other hand, there is no source of oxygen. Oxygen enters the ocean in the surface water through contact with the atmosphere. From there the oxygen is then brought to greater depths through the sinking and circulation of water masses. These, in turn, are dynamic processes that are strongly affected by climatic conditions. (Chapter 1).
Three factors ultimately determine how high the concentration of dissolved oxygen is at any given point within the ocean:- The initial oxygen concentration that this water possessed at its last contact with the atmosphere.
- The amount of time that has passed since the last contact with the atmosphere. This can, in fact, be decades or centuries.
- Biological oxygen consumption that results during this time due to the respiration of all the consumers. These range from miniscule bacteria to the zooplankton, and up to the higher organisms such as fish.
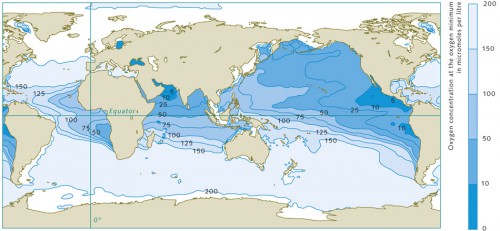
Different groups of marine organisms react to the oxygen deficiency in completely different ways, because of the wide range of tolerance levels of different marine animals to oxygen-poor conditions. For instance, crustaceans and fish generally require higher oxygen concentrations than mussels or snails. The largest oceanic oxygen minimum zones, however, because of their extremely low concentrations, should be viewed primarily as natural dead zones for the higher organisms, and by no means as caused by humans.
- 2.15 > Marine regions with oxygen deficiencies are completely natural. These zones are mainly located in the mid-latitudes on the west sides of the continents. There is very little mixing here of the warm surface waters with the cold deep waters, so not much oxygen penetrates to greater depths. In addition, high bioproductivity and the resulting large amounts of sinking biomass here lead to strong oxygen consumption at depth, especially between 100 and 1000 metres.
Oxygen – the renaissance of a hydrographic parameter
Oxygen distribution in the ocean depends on both biological processes, like the respiration of organisms, and on physical processes such as current flow. Changes in either of these processes should therefore lead to changes in the oxygen distribution. In fact, dissolved oxygen can be viewed as a kind of sensitive early warning system for global (climate) change in the ocean. Scientific studies show that this early warning system can detect the expected decrease in oxygen transport from the atmosphere into the ocean that is driven by global current and mixing processes, as well as possible changes in the marine biotic communities. In recent years, this knowledge has led to a renaissance of oxygen in the field of global marine research.
- In oceanography, dissolved oxygen has been an important measurement parameter for over a hundred years. A method for determining dissolved oxygen was developed as early as the end of the 19th century, and it is still applied in an only slightly modified form today as a precise method. This allowed for the development of an early fundamental understanding of the oxygen distribution in the world ocean, with the help of the famous German Atlantic Expedition of the “Meteor” in the 1920s.
Research efforts in recent years have recorded decreasing oxygen concentrations for almost all the ocean basins. These trends are, in part, fairly weak and mainly limited to water masses in the upper 2000 metres of the ocean. Therefore, no fully consistent picture can yet be drawn from the individual studies. Most of the studies do, however, show a trend of decreasing oxygen concentrations. This trend agrees well with an already verified expansion and intensification of the natural oxygen minimum zones, those areas that are deadly for higher organisms. If the oxygen falls below certain (low) threshold values, the water becomes unsuitable for higher organisms. Sessile, attached organisms die. Furthermore, the oxygen deficiency leads to major changes in biogeochemical reactions and elemental cycles in the ocean – for instance, of the plant nutrients nitrate and phosphate.
- Oxygen levels affect geochemical processes in the sediment but also, above all, bacterial metabolism processes, which, under altered oxygen conditions, can be changed dramatically. It is not fully possible today to predict what consequences these changes will ultimately have. In some cases it is not even possible to say with certainty whether climate change will cause continued warming, or perhaps even local cooling. But it is probable that the resulting noticeable effects will continue over a long time period of hundreds or thousands of years
- Even today, however, climate change is starting to cause alterations in the oxygen content of the ocean that can have negative effects. For the first time in recent years, an extreme low-oxygen situation developed off the coast of Oregon in the United States that led to mass mortality in crabs and fish. This new death zone off Oregon originated in the open ocean and presumably can be attributed to changes in climate. The prevailing winds off the west coast of the USA apparently changed direction and intensity and, as a result, probably altered the ocean currents. Researchers believe that the change caused oxygen-poor water from greater depths to flow to surface waters above the shelf.
The death zone off Oregon is therefore different than the more than 400 near-coastal death zones known worldwide, which are mainly attributed to eutrophication, the excessive input of plant nutrients. Eutrophication normally occurs in coastal waters near densely populated regions with intensive agricultural activity. (Chapter 4)
The Atlantic Expedition For the first time, during the German Atlantic Expedition (1925 to 1927) with the research vessel “Meteor”, an entire ocean was systematically sampled, both in the atmosphere and in the water column. Using an echosounder system that was highly modern for its time, depth profiles were taken across 13 transits of the entire ocean basin.
Oxygen – challenge to marine research
The fact that model calculations examining the effects of climate change almost all predict an oxygen decline in major parts of the ocean, which agrees with the available observations of decreasing oxygen, gives the subject additional weight. Even though the final verdict is not yet in, there are already indications that the gradual loss of oxygen in the world ocean is an issue of great relevance which possibly also has socio-economic repercussions, and which ocean research must urgently address.
Intensified research can provide more robust conclusions about the magnitude of the oxygen decrease. In addition it will contribute significantly to a better understanding of the effects of global climate change on the ocean. In recent years marine research has addressed this topic with increased vigour, and has already established appropriate research programmes and projects. It is difficult, however, to completely measure the temporally and spatially highly variable oceans in their totality. In order to draw reliable conclusions, therefore, the classic instruments of marine research like ships and taking water samples will not suffice. Researchers must begin to apply new observational concepts.
“Deep drifters” are an especially promising tool: these are submersible measuring robots that drift completely autonomously in the ocean for 3 to 4 years, and typically measure the upper 2000 metres of the water column every 10 days. After surfacing, the data are transferred to a data centre by satellite. There are presently around 3200 of these measuring robots deployed for the international research programme ARGO, named after a ship from Greek mythology. Together they form a worldwide autonomous observatory that is operated by almost 30 countries.
So far this observatory is only used on a small scale for oxygen measurements. But there has been developed a new sensor technology for oxygen measurements in the recent past that can be deployed on these drifters. This new technology would give fresh impetus to the collection of data on the variability of the oceanic oxygen distribution.